

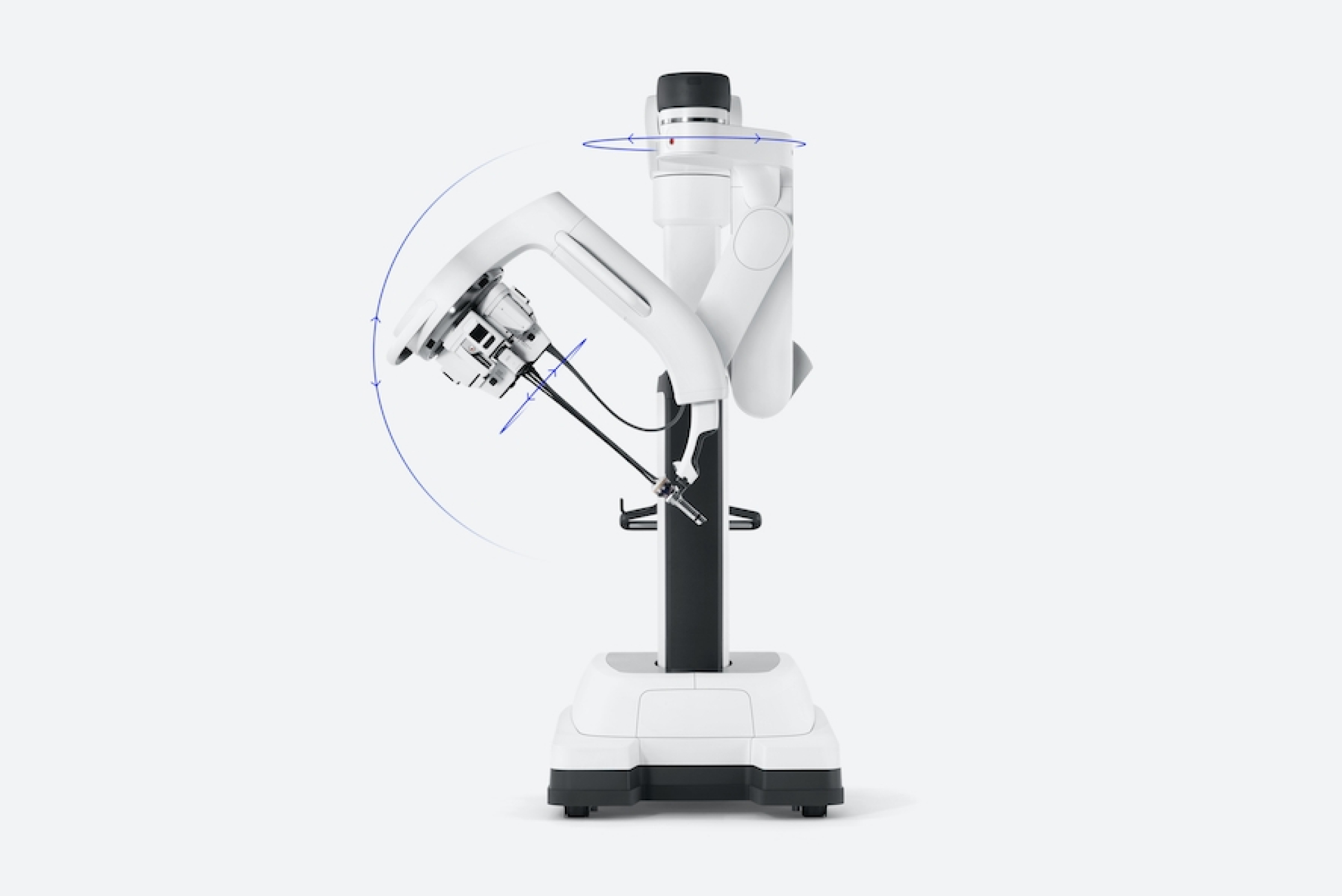
Introducing da Vinci SP by Intuitive, a surgical system engineered to perform robotic-assisted surgery through a single incision or natural orifice.
Da Vinci SP’s unique patient cart design, instruments optimized for single-port surgery, and advanced vision empowers surgeons to pioneer new surgical approaches that preserve healthy tissue and aim to improve patient quality of life.
Since the introduction of da Vinci SP, an emerging body of clinical evidence shows that surgeons are harnessing its unique attributes to move surgery forward, again.
Each da Vinci system offers unique strengths to support a more personalized strategy. Da Vinci SP is designed for precision in compact spaces, including extraperitoneal or natural orifice approaches.
Urology, head and neck, thoracic, and colorectal surgeons are harnessing da Vinci SP to help their patients return to what matters most.

Sign up to receive news about the latest da Vinci SP developments and get started with da Vinci SP for your hospital or practice.
Thank you for your submission.
Important safety information
Serious complications may occur in any surgery, including da Vinci surgery, up to and including death. Serious risks include, but are not limited to, injury to tissues and organs and conversion to other surgical techniques which could result in a longer operative time and/or increased complications. For summary of the risks associated with surgery refer to www.intuitive.com/safety.
For product intended use and/or indications for use, risks, cautions, and warnings and full prescribing information, refer to the associated user manual(s) or visit https://manuals.intuitivesurgical.com/market.
Individual outcomes may depend on a number of factors—including but not limited to—patient characteristics, disease characteristics, and/or surgeon experience.