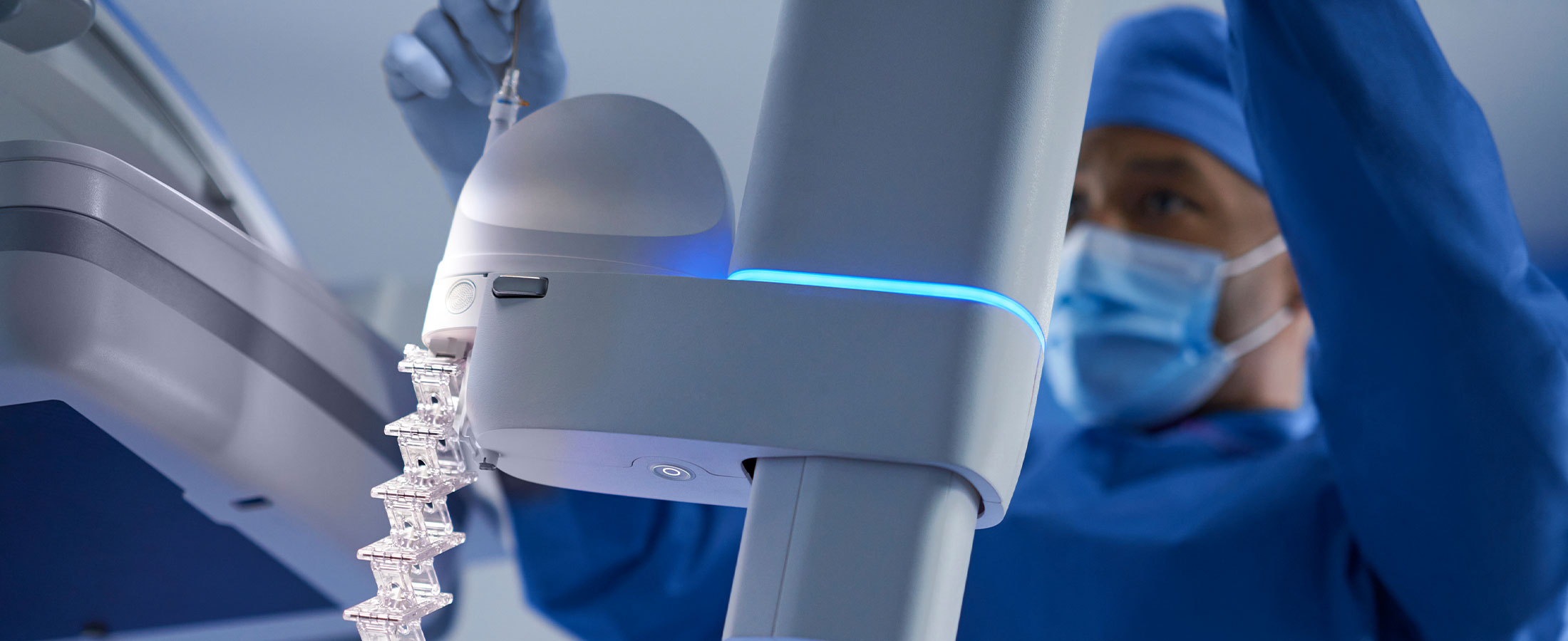

In lung cancer, early detection and diagnosis are critical. Yet today, nearly half of all lung cancer cases are diagnosed at a distant stage, when treatment options are limited.1 Ion delivers an innovative solution for clinicians to get early answers and continue to advance care for lung cancer patients.
We built Ion on the belief that obtaining lung tissue samples for biopsy could be safe and simple, even when lung nodules are small and located in difficult-to-reach locations. Ion’s ultrathin, ultramaneuverable catheter allows clinicians to reach small lesions in all 18 segments of the lung with more reach, precision, and stability.2
Patients who learn they have a suspicious lung nodule shouldn’t have to wait to find out if they have lung cancer. Instead, they should be able to get definitive answers without delay. Yet today, some patients are told to return months later for a follow-up CT scan to see if the nodule has grown3. Patients may also require multiple biopsies prior to a lung cancer diagnosis, which can add months to a patient’s journey to care4 reducing overall survivability and increasing the likelihood of recurrence.5
A chief goal in designing Ion was to help shorten the patient journey by making it possible to biopsy small, difficult-to-reach nodules in the peripheral lung, where more than 70% of cancerous lung nodules may be located.6 Since Ion’s introduction in 2019, it has been reshaping the lung biopsy landscape by allowing physicians to biopsy these challenging nodules.
Visit our Research and Outcomes page to learn what recent studies say about how Ion is helping physicians get early answers for their patients.
At Intuitive, our ongoing commitment to collaborative innovation focuses on providing lung cancer centers with a platform for the future. In partnership with the care community, we continually advance the value of our Ion system, product portfolio, and services to continually advance lung care.
Thank you for your submission.
Important safety information
As with any bronchoscopic procedure under general anesthesia, serious complications may include bleeding, pneumothorax (collapsed lung), cardiac-related complications, respiratory failure, air embolism, or death. For summary of the risks associated with bronchoscopy refer to www.intuitive.com/safety.
For risks, cautions, and warnings and full prescribing information, refer to the associated Ion System user manual(s) or visit https://manuals.intuitivesurgical.com/market.
Individuals’ outcomes may depend on a number of factors, including but not limited to patient characteristics, disease characteristics, and physician experience.
Ion endoluminal system
The Ion endoluminal system (Model IF1000) assists the user in navigating a catheter and endoscopic tools in the pulmonary tract using endoscopic visualization of the tracheobronchial tree for diagnostic and therapeutic procedures. The Ion endoluminal system enables fiducial marker placement. It does not make a diagnosis and is not for pediatric use.
Information provided by the Ion endoluminal system or its components should be considered guidance only and not replace clinical decisions made by a trained physician.
The Flexision Biopsy Needle is used with the Ion Endoluminal System to biopsy tissue from a target area in the lung.
The PlanPoint Software uses patient CT scans to create a 3D plan of the lung and navigation pathways for use with the Ion Endoluminal System.